Wednesday, 16 May 2007
3rd Floor Hall (Pfahler Hall)
68
A novel route to tetra-substituted pyrazoles using CAN
����������� Functionalized pyrazoles are playing an increasingly important role in the development of new pharmaceuticals and have established their place in the agrochemical industry.� The drug celecoxib, antidiabetic agents, antitumor agents, kinase inhibitors, and HIV-1 reverse transcriptase inhibitors all contain pyrazole motifs.� Recently, 4-alkyl-1,3,5-triarylpyrazoles have been examined in detail because of their potential biological activity.� More specifically, propylpyrazole triol (PPT) has been shown to be an equivalent to β-estradiol as a ligand for the estrogen receptor (ER).� In the present work, a method using ceric ammonium nitrate (CAN) for synthesizing PPT as well as other ER ligand candidates has been developed through allylation of β-diketones and subsequent reaction with various substituted hydrazines.� Experimental results show that a wide range of aliphatic and aromatic substituted diketones can be used with this method. ��In unsymmetrical β-diketones, steric hindrance provides the driving force for selectivity.� The total synthesis of PPT using this method provides an overall isolated yield of 30.4%. 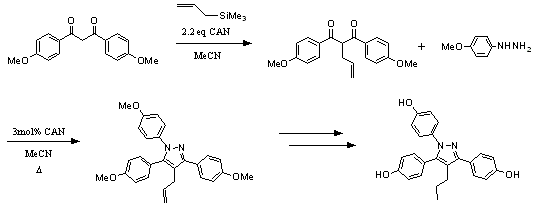
Back to Poster Session I
Back to The Middle Atlantic Regional Meeting (May 16 - 18, 2007)